Homarylamine
Chemical compound
Pharmaceutical compound
 | |
| Clinical data | |
|---|---|
| Other names | 1,3-benzodioxolyl-N-methyl-5-ethanamine; 3,4-methylenedioxy-N-methyl-2-phenylethylamine; Norlobivine |
| Routes of administration | Various |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
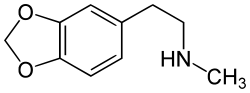
| Formula | C10H13NO2 |
| Molar mass | 179.219 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Homarylamine (INN;[1] also known as 3,4-methylenedioxy-N-methylphenethylamine and MDMPEA) is an antitussive (anti-cough) drug[2] which was patented in 1956 by Merck & Co.,[3] but has never been used medically as such.
Chemically it is a substituted phenethylamine. It is the N-methylated analog of methylenedioxyphenethylamine (MDPEA). It is a schedule I drug in the USA as a positional isomer of MDA.

Reactions
[edit]Reaction of homoarylamine with formaldehyde gives hydrastinine.
A practical application of homarylamine is in the synthesis of Roemerin [de].[4]
See also
[edit]- Lobivine (MDDMPEA)
- Hydrastine, an alkaloid derivative of homarylamine
References
[edit]- ^ "International Non-Proprietary Names for Pharmaceutical Preparations" (PDF). Chronicle of the World Health Organization. 12 (3). 1958.
- ^ Stefko PL, Denzel J, Hickey I (March 1961). "Experimental Investigation of Nine Antitussive Drugs". Journal of Pharmaceutical Sciences. 50 (3): 216–221. Bibcode:1961JPhmS..50..216S. doi:10.1002/jps.2600500309.
- ^ U.S. patent 2,820,739
- ^ Marion, L., Grassie, V. (August 1944). "The Synthesis of l-Roemerine 1". Journal of the American Chemical Society. 66 (8): 1290–1292. Bibcode:1944JAChS..66.1290M. doi:10.1021/ja01236a024.
| DRAsTooltip Dopamine releasing agents |
| ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NRAsTooltip Norepinephrine releasing agents |
| ||||||||||||||
| SRAsTooltip Serotonin releasing agents |
| ||||||||||||||
| Others |
| ||||||||||||||
| Phenethylamines |
| ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amphetamines |
| ||||||||||||||||
| Phentermines |
| ||||||||||||||||
| Cathinones |
| ||||||||||||||||
| Phenylisobutylamines (and further-extended) | |||||||||||||||||
| Catecholamines (and close relatives) |
| ||||||||||||||||
| Cyclized phenethylamines |
| ||||||||||||||||
| Related compounds |
| ||||||||||||||||
This drug article relating to the respiratory system is a stub. You can help Wikipedia by expanding it. |