Glucagon-like peptide-1 receptor
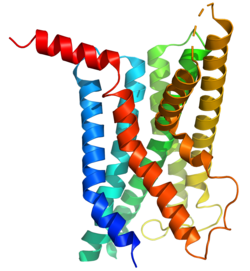
The glucagon-like peptide-1 receptor (GLP1R) is a G protein-coupled receptor (GPCR) found on beta cells of the pancreas and on neurons of the brain. It is involved in the control of blood sugar level by enhancing insulin secretion. In humans it is synthesised by the gene GLP1R, which is present on chromosome 6.[5][6] It is a member of the glucagon receptor family of GPCRs.[7] GLP1R is composed of two domains, one extracellular (ECD) that binds the C-terminal helix of GLP-1,[8] and one transmembrane domain (TMD)[9] that binds the N-terminal region of GLP-1.[10][11][12] In the TMD domain a fulcrum of polar residues regulates the biased signaling of the receptor [10] while the transmembrane helical boundaries[13] and extracellular surface are a trigger for biased agonism.[11]
Ligands
[edit]GLP1R binds glucagon-like peptide-1 (GLP1) and glucagon as its natural endogenous agonists.[14]
- GLP-1 – endogenous in humans[14]
- glucagon – endogenous in humans[14]
- oxyntomodulin
- amycretin
- exendin-4[14][15]
- exenatide
- lixisenatide[14]
- albiglutide
- beinaglutide
- dulaglutide
- efpeglenatide
- langlenatide
- liraglutide[14]
- polyethylene glycol loxenatide
- semaglutide
- taspoglutide
- ecnoglutide
- utreglutide
- glepaglutide
- apraglutide
- maridebart cafraglutide
- tirzepatide
- pegapamodutide
- mazdutide
- survodutide
- bamadutide
- pemvidutide
- cotadutide
- retatrutide
- lithium chloride
- cinchonine
- grutalumab
- dapiglutide
- DA1726
- GX-G6
- GZR18
- HRS9531
- PB718
- RAY1225
- VCT220
- VK2735
- BLX7006
- supaglutide (efsubaglutide)
- ASC30
- danuglipron
- aleniglipron
- lotiglipron
- orforglipron
- CT-996
- CT-388
- CT-868
- HEC88473
- HS-10535
- UBT251
- efinopegdutide
- efocipegtrutide
- NNC9204-1706
- TG103
- Positive:
Structure
[edit]
The GLP-1 receptor is a transmembrane protein composed of seven alpha-helical transmembrane domains (TM1-TM7), an extracellular N-terminus, and an intracellular C-terminus. It belongs to the class B family of G protein-coupled receptors, also known as secretin-like receptors. The extracellular N-terminus contains key regions involved in ligand recognition and binding. It undergoes conformational changes upon ligand binding, leading to activation of intracellular signaling cascades. The intracellular C-terminus interacts with G proteins and other signaling molecules to initiate cellular responses.
Function
[edit]Glucagon-like peptide-1 (GLP-1) is a hormone consisting of 30 amino acids. GLP-1 is released by intestinal L cells when nutrients are consumed. GLP1R is expressed on beta cells in the pancreas. Binding of GLP-1 to GLP1R has multiple effects, including enhancing insulin secretion from pancreatic beta cells in response to glucose, increasing insulin expression, preventing beta-cell apoptosis, promoting the formation of new beta cells, reducing glucagon secretion, slowing down stomach emptying, promoting satiety, and improving glucose disposal in peripheral tissues.
GLP1R is also expressed in the brain[18] where it is involved in the control of appetite.[19]
Mechanism of action
[edit]Upon binding to its ligand GLP-1, the GLP-1 receptor activates intracellular signaling pathways that regulate insulin secretion, glucose metabolism, and satiety.
In pancreatic beta cells, GLP-1 receptor activation enhances glucose-stimulated insulin secretion. This occurs through the activation of adenylyl cyclase, leading to increased intracellular levels of cyclic AMP (cAMP). The rise in cAMP activates protein kinase A (PKA), which promotes insulin exocytosis and enhances beta cell survival and proliferation. GLP-1 receptor signaling in pancreatic alpha cells reduces glucagon secretion, further contributing to glucose lowering.
Activation of GLP-1 receptor delays the rate at which the stomach empties, leading to increased satiety and feeling of fullness.
Activation of the GLP-1 receptor in the brain promotes feelings of satiety.[19]
Clinical significance
[edit]GLP-1 receptor agonists are a class of medications that mimic the actions of the endogenous incretin hormone GLP-1, and are used in type 2 diabetes and obesity.
References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000112164 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000024027 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Thorens B (September 1992). "Expression cloning of the pancreatic beta cell receptor for the gluco-incretin hormone glucagon-like peptide 1". Proceedings of the National Academy of Sciences of the United States of America. 89 (18): 8641–5. Bibcode:1992PNAS...89.8641T. doi:10.1073/pnas.89.18.8641. PMC 49976. PMID 1326760.
- ^ Dillon JS, Tanizawa Y, Wheeler MB, Leng XH, Ligon BB, Rabin DU, et al. (October 1993). "Cloning and functional expression of the human glucagon-like peptide-1 (GLP-1) receptor". Endocrinology. 133 (4): 1907–10. doi:10.1210/endo.133.4.8404634. PMID 8404634.
- ^ Brubaker PL, Drucker DJ (2002). "Structure-function of the glucagon receptor family of G protein-coupled receptors, coupled to: the glucagon, GIP, GLP-1, and GLP-2 receptors" (PDF). Receptors & Channels. 8 (3–4): 179–88. doi:10.1080/10606820213687. PMID 12529935.
- ^ Underwood CR, Garibay P, Knudsen LB, Hastrup S, Peters GH, Rudolph R, Reedtz-Runge S (January 2010). "Crystal structure of glucagon-like peptide-1 in complex with the extracellular domain of the glucagon-like peptide-1 receptor". The Journal of Biological Chemistry. 285 (1): 723–30. doi:10.1074/jbc.M109.033829. PMC 2804221. PMID 19861722.
- ^ Song G, Yang D, Wang Y, de Graaf C, Zhou Q, Jiang S, et al. (June 2017). "Human GLP-1 receptor transmembrane domain structure in complex with allosteric modulators". Nature. 546 (7657): 312–315. Bibcode:2017Natur.546..312S. doi:10.1038/nature22378. PMID 28514449. S2CID 2141649.
- ^ a b Wootten D, Reynolds CA, Koole C, Smith KJ, Mobarec JC, Simms J, et al. (March 2016). "A Hydrogen-Bonded Polar Network in the Core of the Glucagon-Like Peptide-1 Receptor Is a Fulcrum for Biased Agonism: Lessons from Class B Crystal Structures". Molecular Pharmacology. 89 (3): 335–47. doi:10.1124/mol.115.101246. PMC 4767408. PMID 26700562.
- ^ a b Wootten D, Reynolds CA, Smith KJ, Mobarec JC, Koole C, Savage EE, et al. (June 2016). "The Extracellular Surface of the GLP-1 Receptor Is a Molecular Trigger for Biased Agonism". Cell. 165 (7): 1632–1643. doi:10.1016/j.cell.2016.05.023. PMC 4912689. PMID 27315480.
- ^ Yang D, de Graaf C, Yang L, Song G, Dai A, Cai X, et al. (June 2016). "Structural Determinants of Binding the Seven-transmembrane Domain of the Glucagon-like Peptide-1 Receptor (GLP1R)". The Journal of Biological Chemistry. 291 (25): 12991–3004. doi:10.1074/jbc.M116.721977. PMC 4933217. PMID 27059958.
- ^ Wootten D, Reynolds CA, Smith KJ, Mobarec JC, Furness SG, Miller LJ, et al. (October 2016). "Key interactions by conserved polar amino acids located at the transmembrane helical boundaries in Class B GPCRs modulate activation, effector specificity and biased signalling in the glucagon-like peptide-1 receptor". Biochemical Pharmacology. 118: 68–87. doi:10.1016/j.bcp.2016.08.015. PMC 5063953. PMID 27569426.
- ^ a b c d e f Maguire JJ, Davenport AP. "GLP-1 receptor". IUPHAR/BPS Guide to PHARMACOLOGY. International Union of Basic and Clinical Pharmacology. Retrieved 13 September 2015.
- ^ Koole C, Reynolds CA, Mobarec JC, Hick C, Sexton PM, Sakmar TP (April 2017). "Genetically encoded photocross-linkers determine the biological binding site of exendin-4 peptide in the N-terminal domain of the intact human glucagon-like peptide-1 receptor (GLP1R)". The Journal of Biological Chemistry. 292 (17): 7131–7144. doi:10.1074/jbc.M117.779496. PMC 5409479. PMID 28283573.
- ^ Biggs EK, Liang L, Naylor J, Madalli S, Collier R, Coghlan MP, et al. (March 2018). "Development and characterisation of a novel glucagon like peptide-1 receptor antibody". Diabetologia. 61 (3): 711–721. doi:10.1007/s00125-017-4491-0. PMC 5890879. PMID 29119245.
- ^ O'Brien A, Andrews S, Baig AH, Bortolato A, Brown JH, Brown GA, et al. (2019-08-09). "Identification of a novel allosteric GLP–1R antagonist HTL26119 using structure-based drug design". Bioorganic & Medicinal Chemistry Letters. 29 (20): 126611. doi:10.1016/j.bmcl.2019.08.015. PMID 31447084. S2CID 201749908.
- ^ Cork SC, Richards JE, Holt MK, Gribble FM, Reimann F, Trapp S (October 2015). "Distribution and characterisation of Glucagon-like peptide-1 receptor expressing cells in the mouse brain". Molecular Metabolism. 4 (10): 718–31. doi:10.1016/j.molmet.2015.07.008. PMC 4588458. PMID 26500843.
- ^ a b Kinzig KP, D'Alessio DA, Seeley RJ (December 2002). "The diverse roles of specific GLP-1 receptors in the control of food intake and the response to visceral illness". The Journal of Neuroscience. 22 (23): 10470–6. doi:10.1523/JNEUROSCI.22-23-10470.2002. PMC 6758755. PMID 12451146.
Further reading
[edit]- van Eyll B, Lankat-Buttgereit B, Bode HP, Göke R, Göke B (July 1994). "Signal transduction of the GLP-1-receptor cloned from a human insulinoma". FEBS Letters. 348 (1): 7–13. Bibcode:1994FEBSL.348....7V. doi:10.1016/0014-5793(94)00553-2. PMID 7517895. S2CID 9085188.
- Liu C, Sun S, Xie J, Li H, Li T, Wu Q, Zhang Y, Bai X, Wang J, Wang X, Li Z, Wang W (2022). "GLP-1R Agonist Exendin-4 Protects Against Hemorrhagic Transformation Induced by rtPA After Ischemic Stroke via the Wnt/β-Catenin Signaling Pathway". Mol Neurobiol. 59 (6): 3649–3664. doi:10.1007/s12035-022-02811-9. PMC 9148281. PMID 35359227.
- Gromada J, Rorsman P, Dissing S, Wulff BS (October 1995). "Stimulation of cloned human glucagon-like peptide 1 receptor expressed in HEK 293 cells induces cAMP-dependent activation of calcium-induced calcium release". FEBS Letters. 373 (2): 182–6. Bibcode:1995FEBSL.373..182G. doi:10.1016/0014-5793(95)01070-U. PMID 7589461. S2CID 28488846.
- Wei Y, Mojsov S (January 1995). "Tissue-specific expression of the human receptor for glucagon-like peptide-I: brain, heart and pancreatic forms have the same deduced amino acid sequences". FEBS Letters. 358 (3): 219–24. Bibcode:1995FEBSL.358..219W. doi:10.1016/0014-5793(94)01430-9. PMID 7843404. S2CID 44371465.
- Lankat-Buttgereit B, Göke R, Stöckmann F, Jiang J, Fehmann HC, Göke B (1994). "Detection of the human glucagon-like peptide 1(7-36) amide receptor on insulinoma-derived cell membranes". Digestion. 55 (1): 29–33. doi:10.1159/000201119. PMID 8112494.
- Graziano MP, Hey PJ, Borkowski D, Chicchi GG, Strader CD (October 1993). "Cloning and functional expression of a human glucagon-like peptide-1 receptor". Biochemical and Biophysical Research Communications. 196 (1): 141–6. Bibcode:1993BBRC..196..141G. doi:10.1006/bbrc.1993.2226. PMID 8216285.
- Stoffel M, Espinosa R, Le Beau MM, Bell GI (August 1993). "Human glucagon-like peptide-1 receptor gene. Localization to chromosome band 6p21 by fluorescence in situ hybridization and linkage of a highly polymorphic simple tandem repeat DNA polymorphism to other markers on chromosome 6". Diabetes. 42 (8): 1215–8. doi:10.2337/diabetes.42.8.1215. PMID 8392011.
- Dillon JS, Tanizawa Y, Wheeler MB, Leng XH, Ligon BB, Rabin DU, et al. (October 1993). "Cloning and functional expression of the human glucagon-like peptide-1 (GLP-1) receptor". Endocrinology. 133 (4): 1907–10. doi:10.1210/endo.133.4.8404634. PMID 8404634.
- Thorens B, Porret A, Bühler L, Deng SP, Morel P, Widmann C (November 1993). "Cloning and functional expression of the human islet GLP-1 receptor. Demonstration that exendin-4 is an agonist and exendin-(9-39) an antagonist of the receptor". Diabetes. 42 (11): 1678–82. doi:10.2337/diabetes.42.11.1678. PMID 8405712.
- Lankat-Buttgereit B, Göke B (1997). "Cloning and characterization of the 5' flanking sequences (promoter region) of the human GLP-1 receptor gene". Peptides. 18 (5): 617–24. doi:10.1016/S0196-9781(97)00001-6. PMID 9213353. S2CID 29733898.
- Frimurer TM, Bywater RP (June 1999). "Structure of the integral membrane domain of the GLP1 receptor". Proteins. 35 (4): 375–86. doi:10.1002/(SICI)1097-0134(19990601)35:4<375::AID-PROT1>3.0.CO;2-2. PMID 10382665.
- Huypens P, Ling Z, Pipeleers D, Schuit F (August 2000). "Glucagon receptors on human islet cells contribute to glucose competence of insulin release". Diabetologia. 43 (8): 1012–9. doi:10.1007/s001250051484. PMID 10990079.
- Hartley JL, Temple GF, Brasch MA (November 2000). "DNA cloning using in vitro site-specific recombination". Genome Research. 10 (11): 1788–95. doi:10.1101/gr.143000. PMC 310948. PMID 11076863.
- Xiao Q, Jeng W, Wheeler MB (December 2000). "Characterization of glucagon-like peptide-1 receptor-binding determinants". Journal of Molecular Endocrinology. 25 (3): 321–35. doi:10.1677/jme.0.0250321. PMID 11116211.
- Bazarsuren A, Grauschopf U, Wozny M, Reusch D, Hoffmann E, Schaefer W, et al. (May 2002). "In vitro folding, functional characterization, and disulfide pattern of the extracellular domain of human GLP-1 receptor". Biophysical Chemistry. 96 (2–3): 305–18. doi:10.1016/S0301-4622(02)00023-6. PMID 12034449.
- Tokuyama Y, Matsui K, Egashira T, Nozaki O, Ishizuka T, Kanatsuka A (October 2004). "Five missense mutations in glucagon-like peptide 1 receptor gene in Japanese population". Diabetes Research and Clinical Practice. 66 (1): 63–9. doi:10.1016/j.diabres.2004.02.004. PMID 15364163.
- Jorgensen R, Martini L, Schwartz TW, Elling CE (March 2005). "Characterization of glucagon-like peptide-1 receptor beta-arrestin 2 interaction: a high-affinity receptor phenotype". Molecular Endocrinology. 19 (3): 812–23. doi:10.1210/me.2004-0312. PMID 15528268.
- Mahon MJ, Shimada M (January 2005). "Calmodulin interacts with the cytoplasmic tails of the parathyroid hormone 1 receptor and a sub-set of class b G-protein coupled receptors". FEBS Letters. 579 (3): 803–7. Bibcode:2005FEBSL.579..803M. doi:10.1016/j.febslet.2004.12.056. PMID 15670850. S2CID 6471940.
- Graaf C, Donnelly D, Wootten D, Lau J, Sexton PM, Miller LJ, et al. (October 2016). "Glucagon-Like Peptide-1 and Its Class B G Protein-Coupled Receptors: A Long March to Therapeutic Successes". Pharmacological Reviews. 68 (4): 954–1013. doi:10.1124/pr.115.011395. PMC 5050443. PMID 27630114.
- Song G, Yang D, Wang Y, de Graaf C, Zhou Q, Jiang S, et al. (June 2017). "Human GLP-1 receptor transmembrane domain structure in complex with allosteric modulators". Nature. 546 (7657): 312–315. Bibcode:2017Natur.546..312S. doi:10.1038/nature22378. PMID 28514449. S2CID 2141649.
- Brunton S (May 2014). "GLP-1 receptor agonists vs. DPP-4 inhibitors for type 2 diabetes: is one approach more successful or preferable than the other?". International Journal of Clinical Practice. 68 (5): 557–567. doi:10.1111/ijcp.12361. PMC 4238422. PMID 24499291.
- Donnelly D (May 2012). "The structure and function of the glucagon-like peptide-1 receptor and its ligands". British Journal of Pharmacology. 166 (1): 27–41. doi:10.1111/j.1476-5381.2011.01687.x. PMC 3415635. PMID 21950636.
External links
[edit]- "Glucagon Receptor Family: GLP-1". IUPHAR Database of Receptors and Ion Channels. International Union of Basic and Clinical Pharmacology. Archived from the original on 2016-03-03. Retrieved 2007-10-25.
- glucagon-like+peptide+receptor at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Overview of all the structural information available in the PDB for UniProt: P43220 (Glucagon-like peptide 1 receptor) at the PDBe-KB.